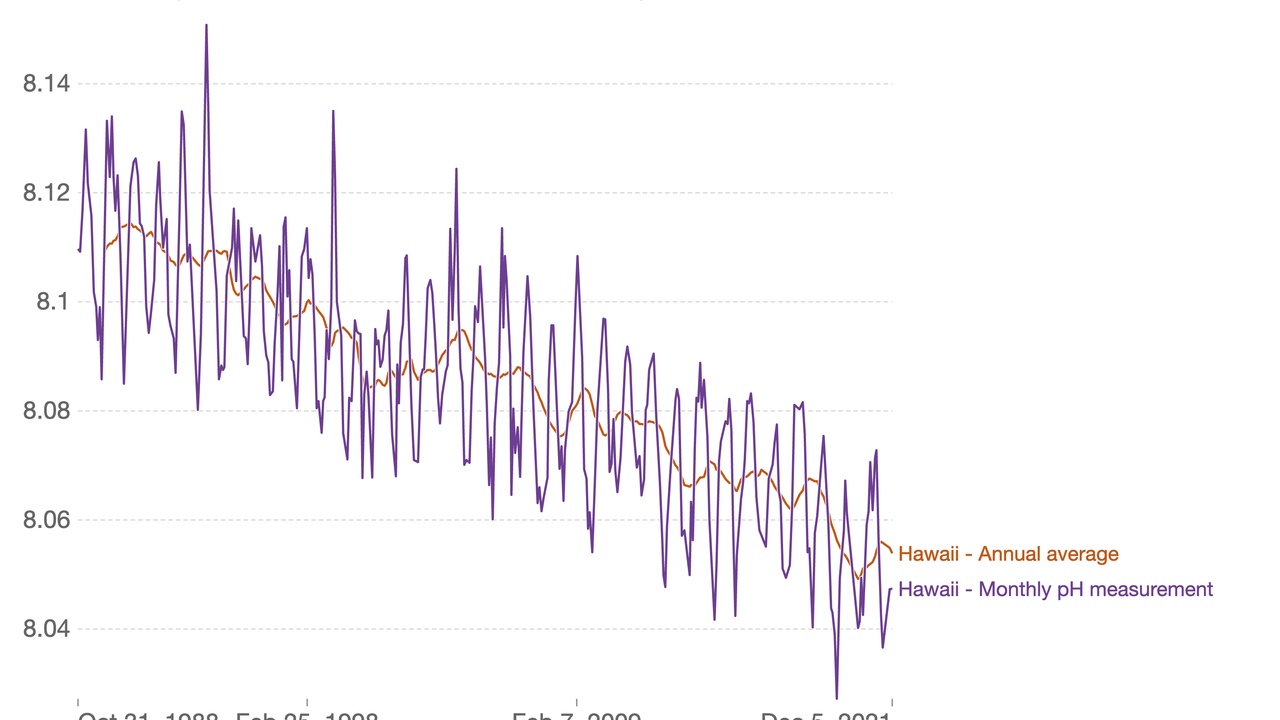
Since the Industrial Revolution (around 1750), surface ocean pH has fallen by about 0.1 units — a change that corresponds to roughly a 30% increase in acidity (hydrogen ion concentration).
That drop happened because seawater absorbs a large share of the CO2 humans release; dissolved CO2 forms carbonic acid and shifts seawater chemistry toward lower pH and fewer carbonate ions, the building blocks many marine animals need for shells and skeletons.
These eight facts about ocean acidification explain how human carbon emissions are reshaping seawater chemistry, why that matters for shell-forming species, fisheries, coral reefs, and coastal economies, and what practical steps can reduce harm.
The science behind ocean acidification
Ocean chemistry shifts when seawater takes up CO2 from the atmosphere. That gas reacts with water to form carbonic acid, which releases hydrogen ions and reduces carbonate ion availability — a chain reaction with predictable chemical outcomes that scientists measure directly in labs and at sea.
Below are three concise scientific facts that explain how acidification works, how fast it has changed, and why conditions differ from place to place.
1. Ocean acidification is a chemical consequence of CO2 emissions
When the ocean absorbs anthropogenic CO2, much of it converts into carbonic acid, lowering pH and shifting the balance of dissolved carbonate species. Observational programs show oceans have taken up roughly 30% of human CO2 emissions since preindustrial times, and surface pH has declined by about 0.1 units since 1750.
Those trends come from long-term surface pH and carbonate monitoring (for example, NOAA surface pH stations and multi-decade shipboard surveys) and from IPCC assessments that quantify ocean CO2 uptake for climate models and budgets.
2. Small pH changes mean large shifts in carbonate chemistry
pH is a logarithmic scale, so a 0.1-unit drop corresponds to roughly a 25–30% increase in hydrogen ion concentration; chemically, that is a substantial change even if it sounds small on the scale.
That extra acidity reduces the concentration of carbonate ions, which are essential for organisms that make calcium carbonate shells or skeletons. Laboratory incubation studies commonly report 10–40% declines in calcification rates under elevated CO2, and field observations show species like pteropods developing thinner or dissolving shells in parts of the North Pacific and Arctic.
3. Acidification varies by region, season, and depth
Acidification is not uniform: coastal upwelling brings deep, CO2-rich waters to the surface, freshwater inputs alter alkalinity near river mouths, and polar seas warm and acidify faster than some temperate regions.
For example, seasonal upwelling off the U.S. West Coast produces low-pH events that have been measured repeatedly off Oregon and California, and Arctic shelf waters are projected to reach corrosive conditions for some organisms earlier this century than open-ocean areas, affecting local monitoring and fisheries management.
Impacts on marine life and ecosystems

Changes in carbonate chemistry affect organisms from microscopic plankton to reef-building corals, and those biological responses cascade through food webs. Below are three facts about direct physiological impacts, food-web consequences, and reef vulnerability, with real-world examples and study findings.
4. Shell-forming species show reduced calcification and survival
Mollusks, pteropods, and many juvenile stages of marine animals often produce thinner shells and experience higher mortality when pH and carbonate saturation fall. Hatchery and lab studies show significant declines in larval survival and calcification under elevated CO2 conditions.
Real-world consequences were apparent in the Pacific Northwest during the 2000s, when corrosive upwelled water contributed to widespread oyster spat losses at hatcheries (2005–2010), prompting industry and agencies to invest in monitoring and water-treatment systems.
5. Food webs can shift when base-level plankton are affected
Plankton and larval stages form the base of many marine food webs, so changes to their growth, behavior, or abundance ripple upward to predators. Experiments and field work indicate copepods, krill, and other zooplankton can show physiological stress and altered timing under acidified conditions.
That can change prey availability for commercially important fish and marine mammals, affecting fish recruitment and the productivity managers rely on to set harvest limits in the North Atlantic and Pacific.
6. Coral reefs face weakened skeletons and slower recovery
Reduced carbonate availability lowers coral calcification rates, weakening reef skeletons and slowing reef accretion. Models project slower reef growth under high CO2 scenarios, and the effect combines with warming-driven bleaching to limit recovery after disturbances.
On the Great Barrier Reef, repeated bleaching events since the late 1990s have reduced coral cover, and acidification makes rebuilding reef structure harder—threatening biodiversity, tourism revenue, and the coastal protection reefs provide.
Human consequences and practical responses

Biological impacts translate into economic and cultural effects for fisheries, aquaculture, coastal protection, and Indigenous communities that rely on marine resources. Below are two facts about the human stakes and actions underway to reduce harm.
7. Ocean acidification has measurable economic and cultural impacts
Declines in shellfish production and altered fish recruitment carry real costs. Regional shellfish aquaculture is worth hundreds of millions of dollars in some areas, and events like the Pacific Northwest oyster spat losses forced producers to absorb lost revenue and invest in mitigation.
Communities that depend on shellfish for subsistence or cultural practices also face losses, prompting local governments and agencies (for example, NOAA and regional partners) to fund monitoring, hatchery upgrades, and research into resilient strains and practices.
8. Monitoring, mitigation, and local adaptation can reduce harm
Cutting CO2 emissions is the essential long-term solution, but local actions already make a difference. Hatcheries that monitor seawater and use buffering (adding alkalinity or lime to incoming seawater) have improved larval survival, and coastal monitoring networks help managers anticipate low-pH events.
Programs such as the Oregon Coastal Acidification Program provide real-time data for industry and managers, while restoration of mangroves and seagrass (blue carbon projects) can enhance local alkalinity and store carbon. Combining emission reductions with monitoring, hatchery buffering, and habitat restoration builds resilience.
Summary
- Oceans have absorbed about 30% of human CO2 since preindustrial times, driving a ~0.1 pH-unit decline at the surface and large chemical shifts that harm calcifiers.
- Small drops in pH mean big changes in carbonate availability, affecting shell-formers from pteropods to oysters and slowing coral reef growth.
- Impacts are regional and seasonal: coastal upwelling, polar amplification, and freshwater inputs create hotspots that need local monitoring and tailored management.
- Practical responses—hatchery buffering, monitoring networks (e.g., Oregon’s program), and blue carbon habitat restoration—can reduce near-term harm while global CO2 cuts address the root cause.
- Support for continued monitoring, science-based policy, and low-carbon choices will help protect fisheries, reefs, and coastal communities from the worst outcomes.